- Press Office
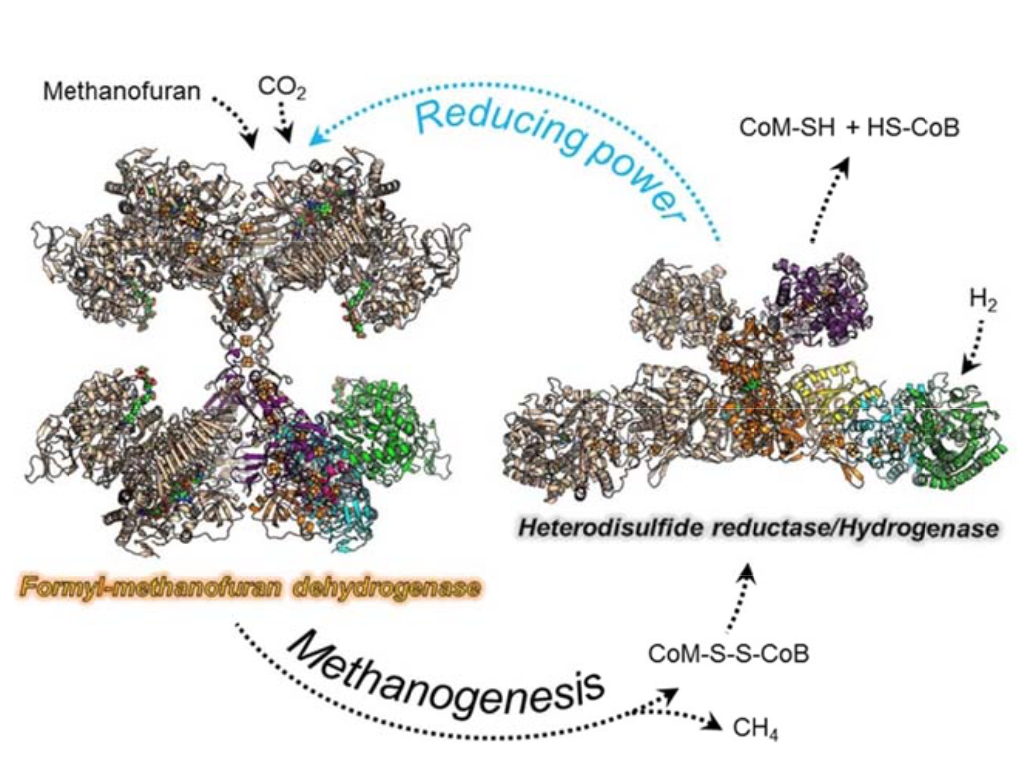
- Dr. Tristan Wagner: "A new way to fix CO2 without ATP...
Dr. Tristan Wagner: "A new way to fix CO2 without ATP consumption, a lesson from methanogenic archaea"

Trace metals like iron (Fe), copper (Cu) and Manganese (Mn), are essential for all aquatic organisms, yet under ambient oxic seawater conditions their solubility is very low. This is why we are interested in reactive oxygen species species like the Superoxide-radical (O2-) or Hydrogen Peroxide (H2O2) as they are known to assess changes in the speciation and cycle metals between thermodynamically favored, but poorly soluble and more bioavailable kinetically favored reduced forms.
Our work on the redox speciation and concentrations of both dissolved and particulate forms of Fe in the Peruvian oxygen minimum zone (OMZ), where oxygen is found to be undetectable, suggests, that dissolved Fe is also here lost by oxidation. The remarkable similarity in the distributions of total particulate Fe and nitrite proposes a role for nitrite in the oxidation. We present a conceptual model for the rapid redox cycling of Fe that occurs in OMZs, despite the absence of oxygen.