- Departments
- Department of Molecular Ecology
- Coastal to Open Ocean Waters
Coastal to Open Ocean Waters

Spring blooms at Helgoland
Every year in spring, with increasing sun light, diatoms and other microalgae start to grow in the North Sea. They rapidly form blooms that are visible from space and are of global importance for carbon cycling. Large amounts of CO2 are removed from the atmosphere and fixed in algal biomass, mostly as polysaccharides and proteins.
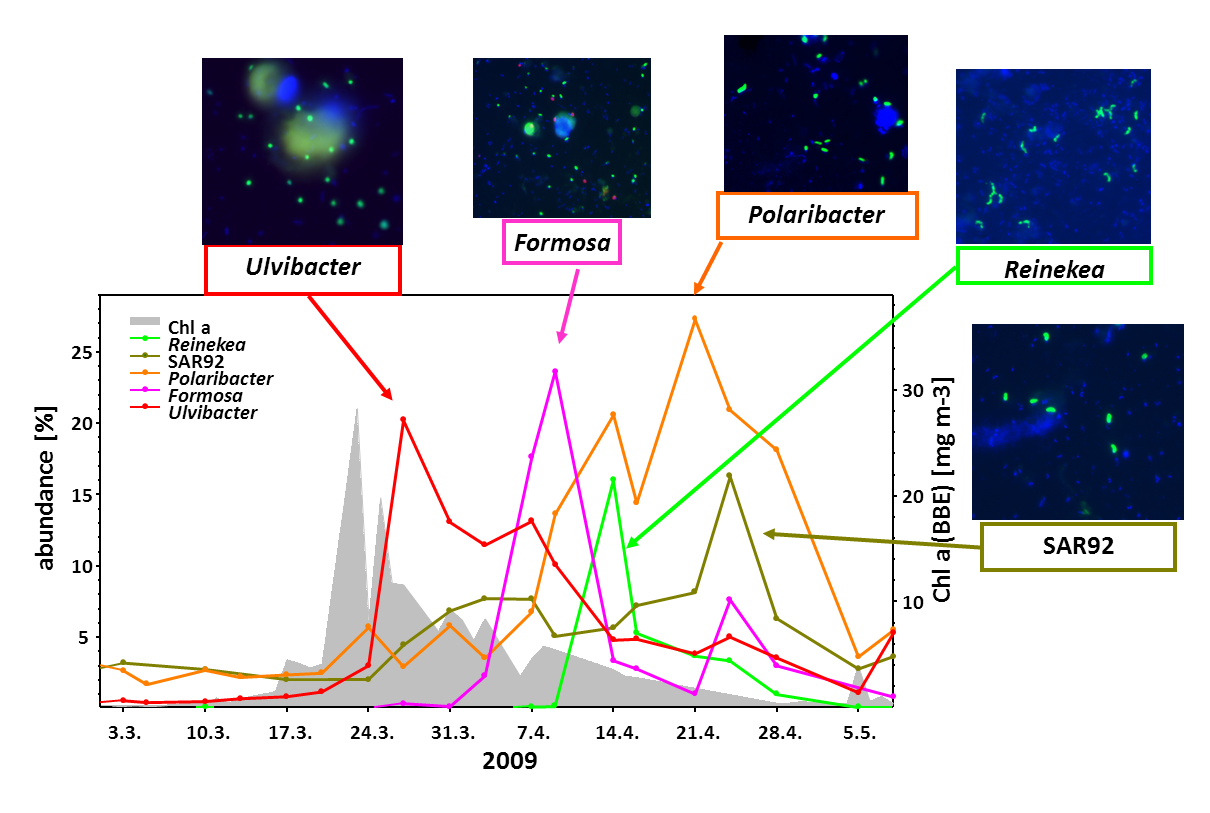
Within few weeks the algal blooms become nutrient limited and eventually break down due to grazing and viral attack. How do which marine bacteria remineralize this algal biomass, thereby releasing most of the CO2? This important research question was explored in great detail over the last years. In a data-rich study (Teeling et al., 2016) we could show that in four subsequent years the same bacterial genera profit from the algal biomass.
Polysaccharide Utillization Loci
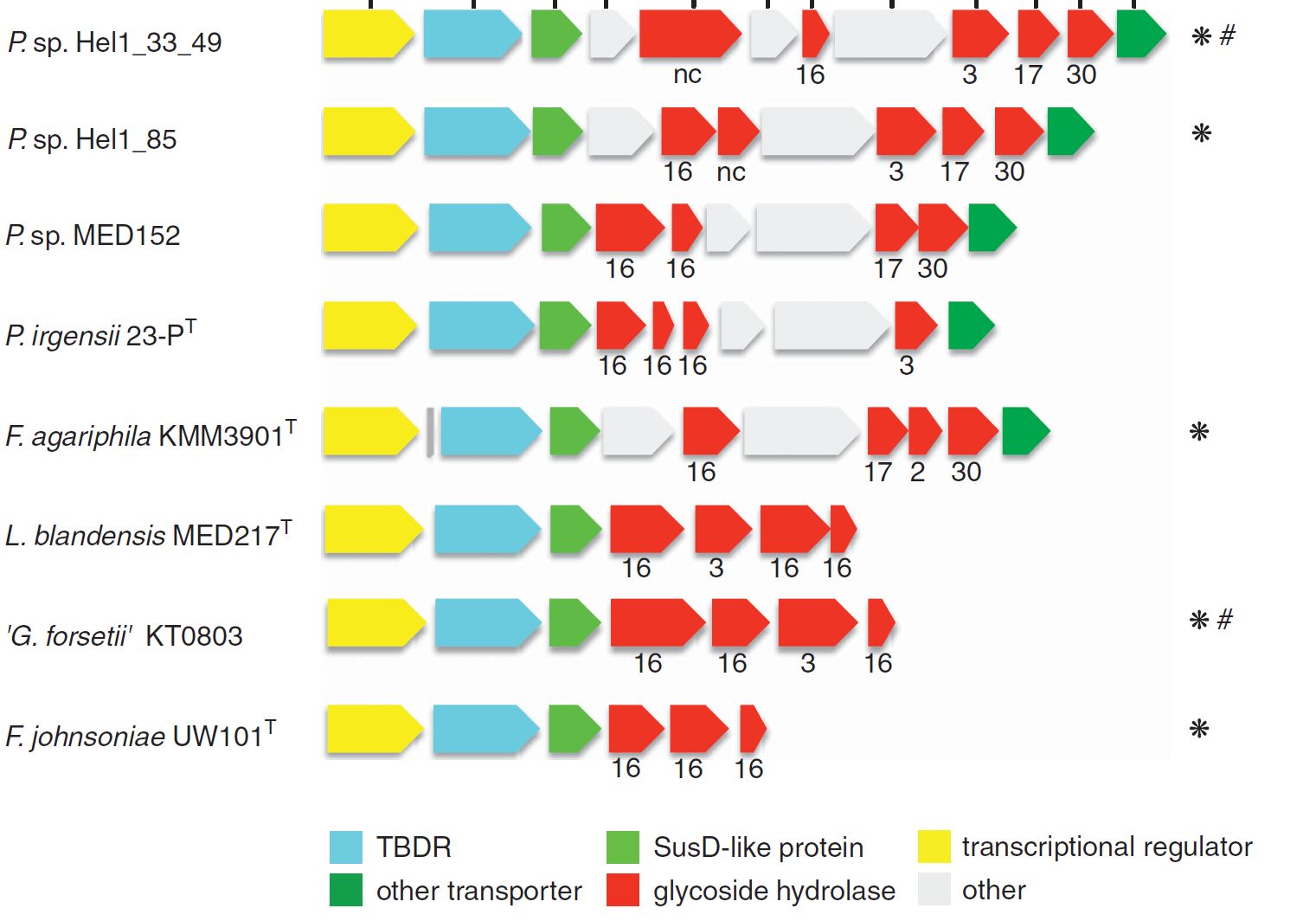
Large operons encoding carbohydrate active enzymes (CAZymes), so-called polysaccharide utilization loci (PULs) are not only a hallmark of gut Bacteroidetes but are also key to the substrate specialization of marine Flavobacteriia.
Last year we published PULs retrieved from contrasting surface waters of the North Atlantic (Bennke et al. 2016), and are currently analyzing 44 metagenomes obtained from the spring blooms 2009-2012 (Krüger et al. unpublished). Additionally about 200 PULs have been identified in the genomes of 60 flavobacterial isolates obtained from the North Sea by the group of Jens Harder (Kappelmann et al. unpublished).
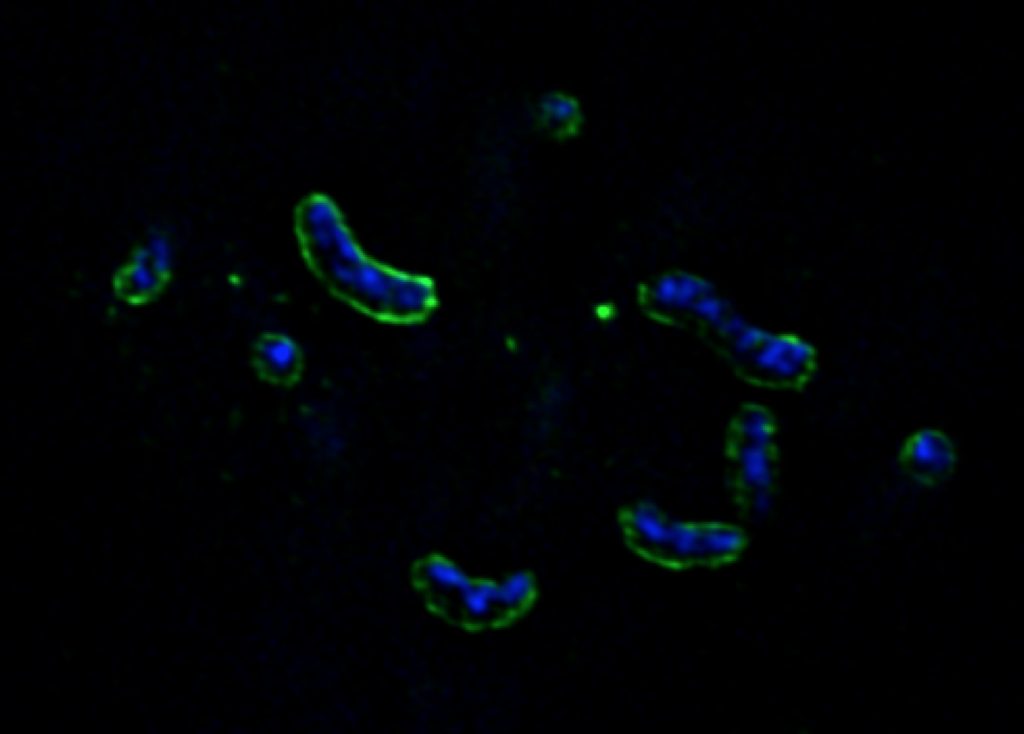
Selfish Bacteria
By serendipity we discovered that the utilization of fluorescently labeled polysaccharides can be visualized by light microcopy (Reintjes et al., 2017). Flavobacteriia are binding and partially degrading polysaccharides at the outer membrane, from where large, still fluorescein-labeled oligosaccharides are rapidly taken up into the periplasmic space via TonB-dependent SusC transporters (see right). Only in this diffusion-protected space the degradation proceeds to mono- and disaccharides which are ultimately transported over the inner membrane into the cytoplasm. This mechanism has been referred to as “selfish” polysaccharide utilization since it avoids the loss of CAZyme and the “sharing” of degradation products with “cheating” bacteria.
Aggregates
Surface waters are characterized by a continuum of organic matter ranging from dissolved molecules to particles, which can form visible marine aggregates. Such particles constitute hot spots of microbial life. We tested the hypothesis that the microbial communities in the 0.2-3 µm size fraction enriched for free-living bacteria would profoundly differ from those collected on filters of 3, 5 or 10 µm pore size which reflect mostly particle-associated microorganisms. Two rRNA-based taxonomic studies supporting this hypothesis have been finished in the reporting period. They were performed in coastal marine and limnic waters (Bizic-Ionescu et al., 2014) and in the West-African Upwelling area (Thiele et al., 2015), and precede future studies in which we plan to investigate the genomic repertoire, in particular the PUL spectrum of free-living and particle associated bacteria.