- Press Office
- Press releases
- How diatoms use chains of sugar to keep bacteria close & friendly
How diatoms use chains of sugar to keep bacteria close & friendly
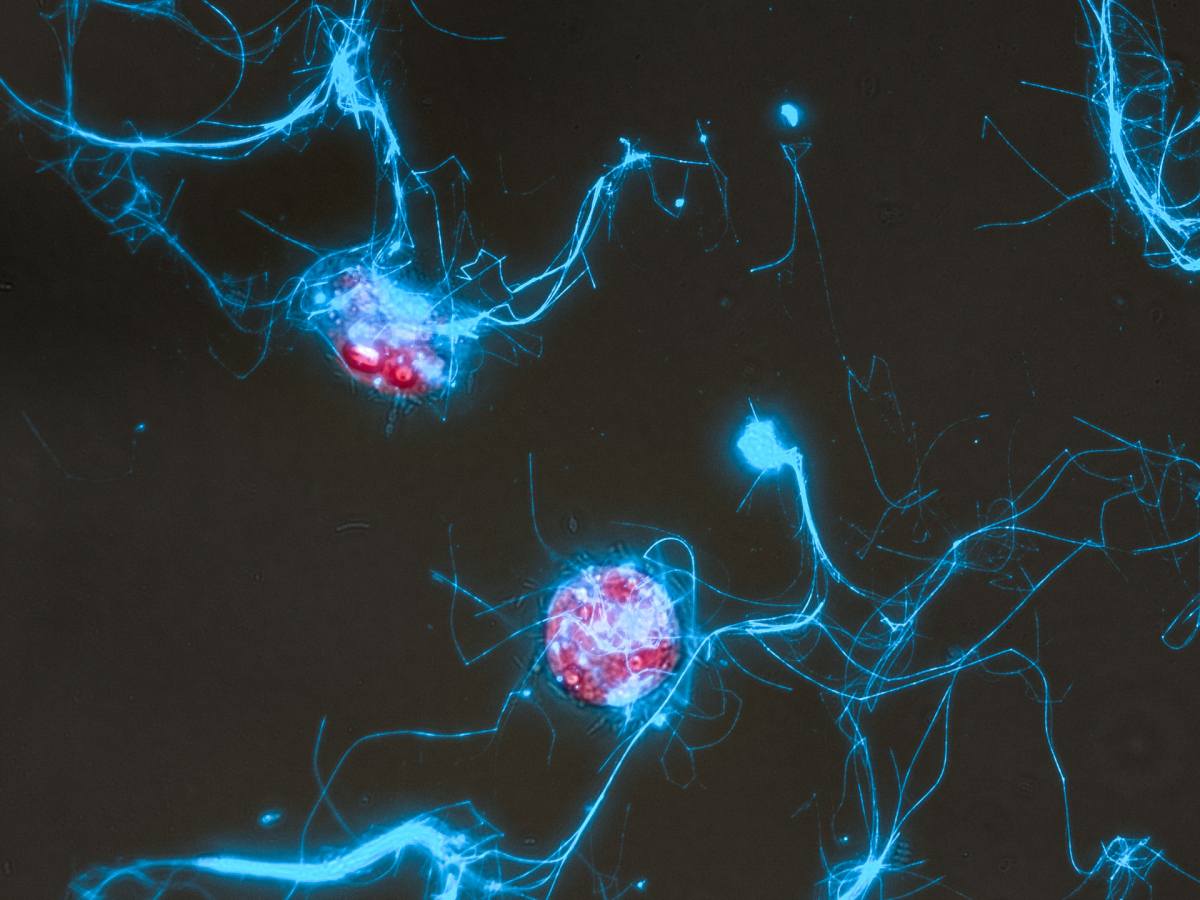
Every spring, when light returns to temperate seas, microscopic algae called diatoms explode in number. These so-called algal blooms are among the most productive biological events on Earth, fixing carbon dioxide and fuelling marine food webs worldwide. But diatoms do not live alone. They host a microbiome, this means they are surrounded by bacteria. The composition of that bacterial community is not random. Now, a team of chemists, microbiologists and ecologists have discovered one of the molecular signals that diatoms use to select their microbiome.
The key is a sugar. A sulfated, negatively charged polysaccharide secreted by diatoms selectively feeds only the bacteria equipped to break it down.
A hidden signal in the water
Diatoms release a large fraction of their photosynthetically fixed carbon into the surrounding seawater as dissolved organic matter, much of it in the form of carbohydrates such as sugars. Researchers have long suspected that some of these sugars might shape which bacteria thrive around diatoms, but the specific molecules involved were unknown.
"We kept seeing the same bacteria turn up wherever diatoms bloomed," says Jan-Hendrik Hehemann from the MARUM - Center for Marine Environmental Sciences at the University of Bremen. "Something had to be connecting them."
A team led by Joris Krull at the Max Planck Institute for Marine Microbiology in Bremen now discovered that the centric diatom Conticribra weissflogii secretes a previously unknown sugar polymer: a structurally regular, highly charged polysaccharide composed almost entirely of mannose, with a sulfate group attached to every sugar unit. Its structure was predicted using nuclear magnetic resonance spectroscopy and confirmed by comparison to a chemically synthesised reference standard.
A sugar only specialists can eat
Not just any bacterium can eat this sulfated mannan. Only bacteria carrying a dedicated set of enzymes, essentially a toolkit specific for this one substrate are able to break it down and use it as a carbon source.
To confirm the predicted structure, the team synthesised reference compounds, short sugar chains, called oligosaccharides. "The structural simplicity of this glycan was surprising," says Conor Crawford from Trinity College Dublin. "Many algal sulfated polysaccharides are too complex to characterise precisely. This one was not."
Joris Krull led the team to identify exactly how the four enzymes worked together. Finding that two enzymes work outside the bacterial cell to break the polymer into smaller pieces, and then two inside finish the job, releasing the simple sugar, mannose.
The authors showed that bacteria lacking this enzymatic toolkit cannot consume the polysaccharide at all, while those carrying it can grow on it as a sole carbon source. The enzymes discovered in this work include an enzyme class not previously known to science.
The same partnership, ocean-wide
Instrumental to this discovery was the long-term microbial and molecular time series dataset collected near the island of Helgoland in the North Sea. This work was possible only through extensive collaboration between technicians, engineers, administrators and scientists hosted at the Max Planck Institute for Marine Microbiology, the University of Greifswald, the MARUM - Center for Marine Environmental Sciences at the University of Bremen and the Alfred Wegener Institute, Helmholtz Centre for Polar and Marine Research (AWI). Successful science requires a village.
The bacteria best equipped to eat this glycan are species of Polaribacter, a group of marine bacteria. “The dataset shows that Polaribacter populations carrying the specific enzymes rise and fall in synchrony with diatom blooms, year after year across many years of observation”, says Chandni Sidhu, who led the metagenomic analysis at the Max Planck Institute for Marine Microbiology.
"What struck us was how consistent it was," says Joris Krull. "The same bacteria, with the same enzymes regularly follow the same diatoms, almost every year.”
To test whether this relationship extends beyond the North Sea, the team searched a global ocean dataset collected by the Tara Oceans expedition, spanning 45 sites across the Atlantic, Pacific and Indian Oceans. They found the genes for mannan degradation were found around the globe. Gene expression data confirmed these enzymes are not only present but actively used in the open ocean.
"We can see these bacterial genes reoccurring across the ocean over time," says Chandni Sidhu. "That tells you this is not a niche curiosity. It is happening out there at a global scale."
The same mannan structure was also found in the secretions of three other ecologically important diatom species, suggesting this is a widespread feature of diatom biology.
An insidious meal
The sulfated mannan appears to do more than simply nourish bacteria. The scientists think its strong negative charge and chemical structure allow it to interact with proteins on bacterial cell surfaces, potentially interfering with bacteria that lack the means to degrade it, while rewarding those that can.
"The diatom is not passively leaking sugar," says Hehemann, “like all eukaryotes surrounded by bacteria they are constantly under pressure to survive. I think they use the polyelectrolyte molecule as an electric fence, a highly charged molecular chain poised to constrain bacteria. This leaves the “friendly” bacteria with no other choice than to eat the fence or die". This principle may be applicable to other microbial consortia e.g. in our intestines exposed to charged polysaccharides such as the ones we consume in fruits and vegetables.
Altogether, the paper defines a chemically specific partnership between diatoms and specialised bacteria, mediated by a single sulfated polysaccharide and a dedicated degradation pathway.
At the scale of ocean blooms, such partnerships may shape not just who eats what, but how carbon is cycled and how much of it ultimately sinks to the deep.
Original publication
Krull, C. Sidhu, V. Solanki, M. Bligh, L. Rößler, R.K. Singh, G. Huang, C.S. Robb, H. Teeling, P.H. Seeberger, T. Schweder, C.J. Crawford & J.-H. Hehemann. Sulfated mannan of diatoms selects host-specific microbiota in the sunlit ocean. Microbiome (2026). https://doi.org/10.1186/s40168-026-02379-9
Participating institutions
- Max Planck Institute for Marine Microbiology, Bremen, Germany
- MARUM Center for Marine Environmental Sciences, University of Bremen, Bremen, Germany
- Max Planck Institute of Colloids and Interfaces, Potsdam, Germany
- School of Chemistry, Trinity Biomedical Sciences Institute, Trinity College Dublin, Dublin, Ireland
- Institute of Pharmacy, University of Greifswald, Greifswald, Germany
Please direct your queries to:
Guest
Department of Molecular Ecology
MPI for Marine Microbiology
Celsiusstr. 1
D-28359 Bremen
Germany

Head of Press & Communications
MPI for Marine Microbiology
Celsiusstr. 1
D-28359 Bremen
Germany
|
Room: |
1345 |
|
Phone: |